View before and after photos IN TEENS
to see results

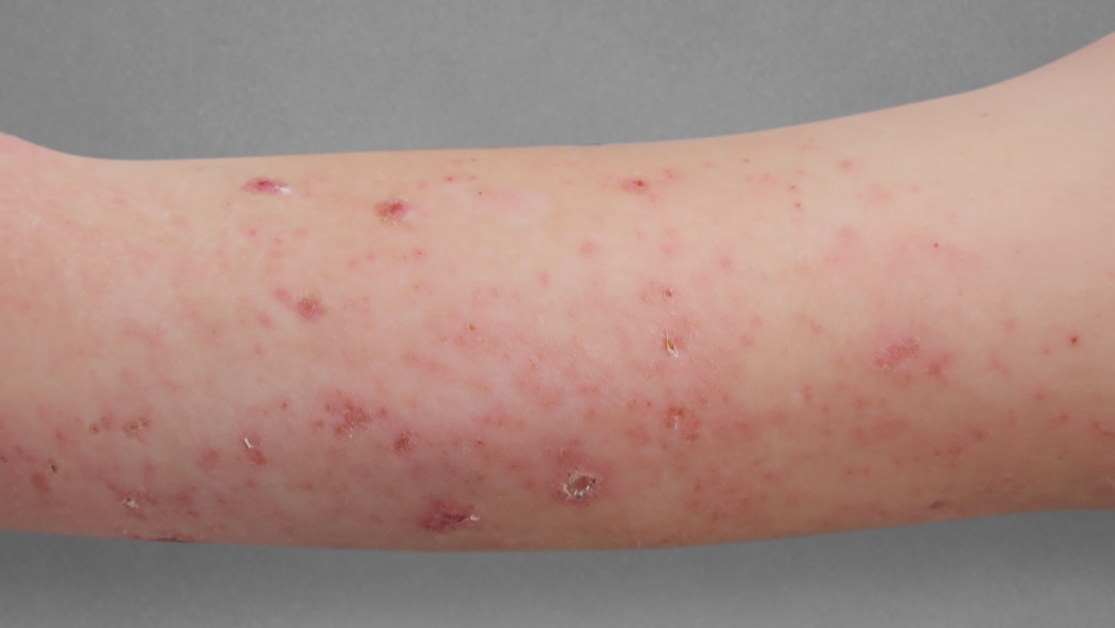
Before Treatment
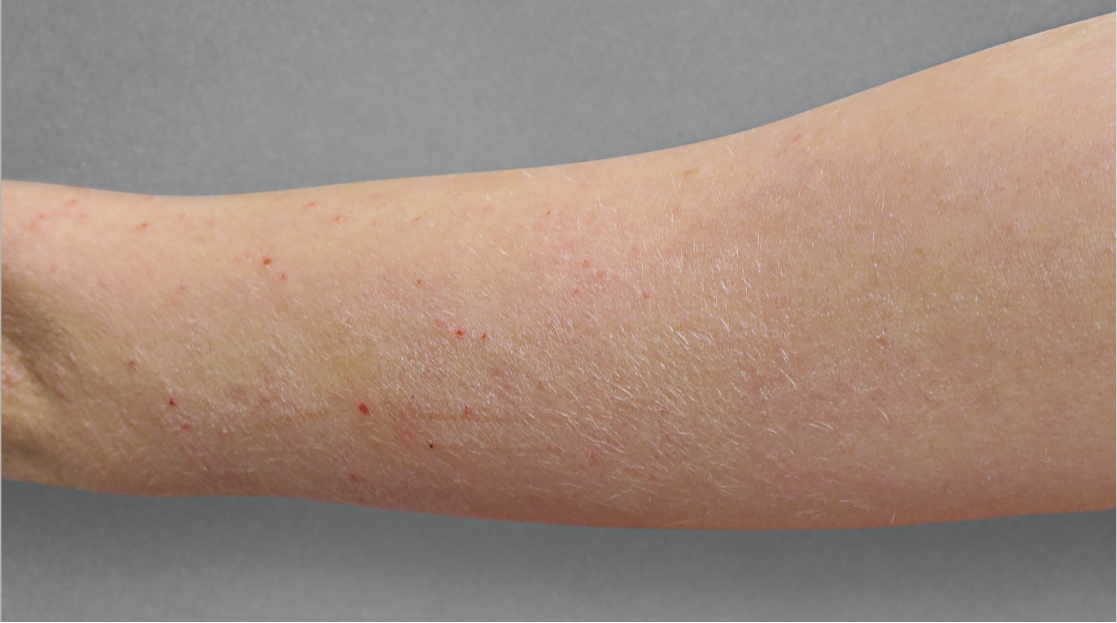
After Treatment with
dupixent at 16 weeks
Actual 12-year-old clinical trial patient treated with DUPIXENT. This patient did not meet clinical trial criteria for clear or almost clear skin at 16 weeks. Individual results may vary.
A proactive treatment for teens
Results from a clinical trial of patients ages 12-17 years with moderate-to-severe eczema at 16 weeks:
ALMOST CLEAR SKIN
24% on DUPIXENT vs
2% not on DUPIXENT
itch relief
37% on DUPIXENT vs
5% not on DUPIXENT
nearly half of
teens
42% on DUPIXENT vs 8% not on DUPIXENT
for some
23% on DUPIXENT vs 2% not on DUPIXENT
LONG-TERM
SAFETY
RESULTS IN teens
Long-term safety results for teens continuing on DUPIXENT in the pediatric extension study through 1 year were consistent with the adult clinical trials.
Most common side
effects include:
- Injection site reactions
- Eye problems, including eye and eyelid inflammation, redness, swelling, itching, eye infection, dry eye, and blurred vision
- Cold sores in your mouth or on your lips
- High count of a certain white blood cell (eosinophilia)
View the possible side effects of DUPIXENT
What you should know about the patients in this study:
atopic dermatitis
atopic dermatitis
surface
area
involvement
Have you spoken to a dermatologist or allergist about DUPIXENT?
Great! Sign up for DUPIXENT MyWay®, our patient support program, to access program benefits and get one-on-one support every step of the way, including help with filling your first prescription.
If you're still struggling with moderate-to-severe eczema, maybe it's time to change the conversation.
Answer a few short questions about your eczema symptoms and treatment goals. We’ll turn it into a personalized guide you can use at your next visit.
Specialists like dermatologists or allergists often have more experience treating moderate-to-severe eczema and can help create a personalized care plan for you.
Stay connected
Feel like you’re struggling with your symptoms?
Sign up for information that can help.